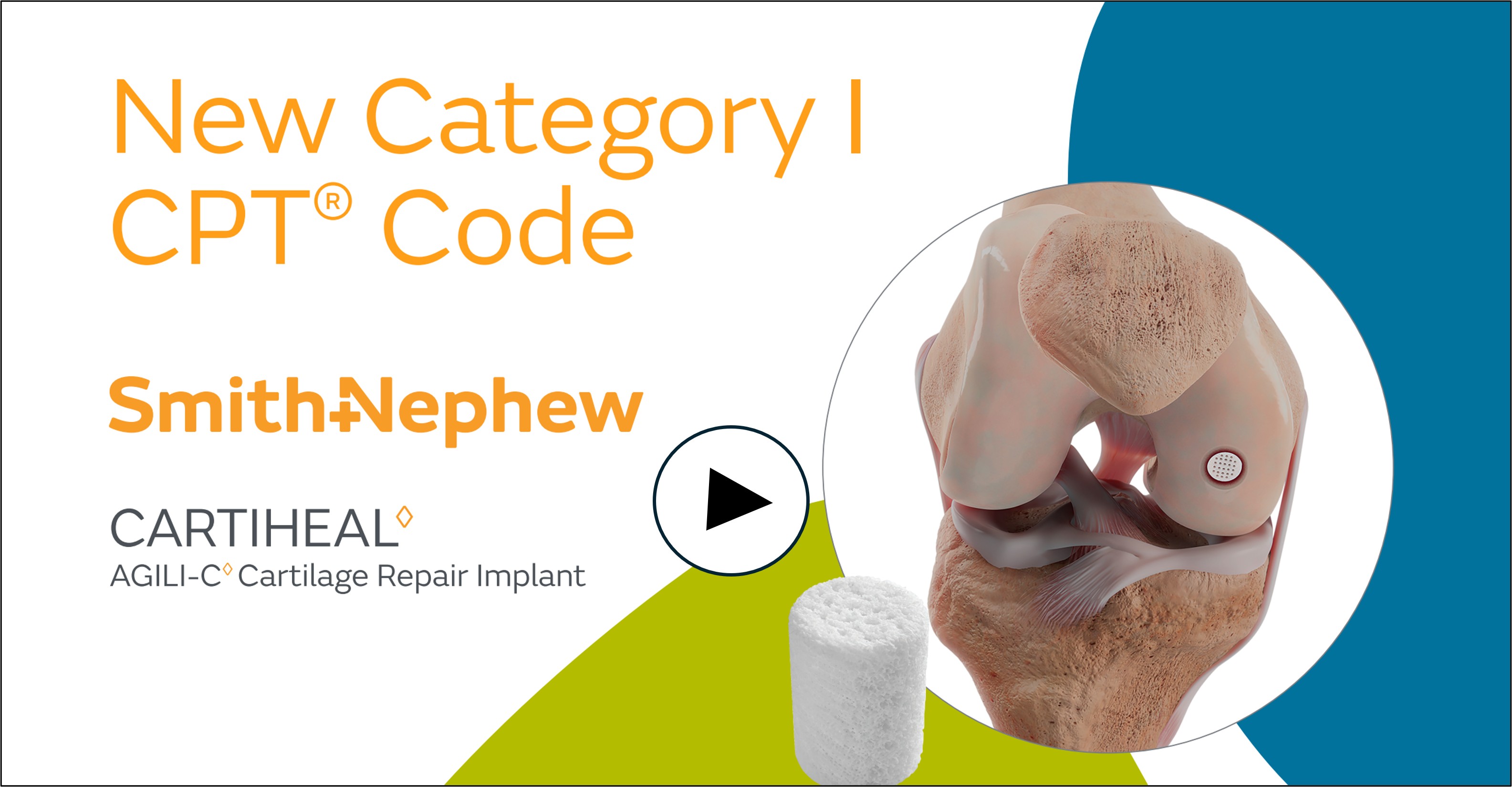
Smith+Nephew Announces New Category I CPT Code for CARTIHEAL AGILI-C Cartilage Repair Implant
Smith+Nephew (LSE:SN, NYSE:SNN), the global medical technology company, today announces that the American Medical Association (AMA) CPT Editorial Panel has established a Category I Current Procedural Terminology (CPT) code for procedures involving its CARTIHEAL AGILI-C Cartilage Repair Implant, effective January 1, 2027.
The new CPT code recognizes the clinical significance and growing adoption of the CARTIHEAL Implant, a single-stage, off-the-shelf solution for treating cartilage and osteochondral defects in the knee, including in patients with mild to moderate osteoarthritis (OA). The implant received Breakthrough Device Designation from the U.S. Food and Drug Administration and is the only FDA-approved device for this indication.
Clinical Impact
The CARTIHEAL Implant demonstrated an 87% reduction in the relative risk of total knee arthroplasty or osteotomy at 4 years compared to microfracture or debridement, according to data published from a multicenter randomized controlled trial.1 This positive data and broad knee indication, including OA, provides an additional treatment option for this patient population earlier in their treatment journey.
“This new CPT code is a major milestone for physicians and patients alike,” said Dr. Ken Zaslav of Northwell Health. “It validates the clinical value of the CARTIHEAL Implant and facilitates broader access to a technology that fills a true unmet need in the world of cartilage repair.”
Reimbursement and Access
The Category I CPT code will streamline reimbursement processes for providers and payers, supporting the integration of the CARTIHEAL Implant into standard clinical practice. It also reflects the AMA’s recognition of the procedure’s clinical efficacy, safety, and widespread physician adoption.
“This code represents a critical milestone on our journey to enable access to patients that can benefit from the CARTIHEAL Implant,” said Christie Van Geffen, SVP Global Sports Medicine Marketing for Smith+Nephew.
About the CARTIHEAL AGILI-C Cartilage Repair Implant
The CARTIHEAL Implant is commercially available in the United States, including Puerto Rico. It is composed of aragonite, a naturally occurring calcium carbonate, and acts as a biphasic scaffold that allows it to repair cartilage and restore subchondral bone.2-6 To learn more about Smith+Nephew’s CARTIHEAL AGILI-C Cartilage Repair Implant, visit www.smith-nephew.com/cartiheal.
- ends –
Media Enquiries
Dave Snyder +1 (978) 749-1440
Smith+Nephew
CPT is a registered trademark of the American Medical Association (AMA). All rights reserved.
References
- Conte P, Anzillotti G, Crawford DC, et al. Differential analysis of the impact of lesions' location on clinical and radiological outcomes after the implantation of a novel aragonite-based scaffold to treat knee cartilage defects. Int Orthop. 2024;48(12):3117-3126
- Altschuler N, Zaslav KR, Di Matteo B, et al. Aragonite-Based Scaffold Versus Microfracture and Debridement for the Treatment of Knee Chondral and Osteochondral Lesions: Results of a Multicenter Randomized Controlled Trial. Am J Sports Med. 2023;51(4):957-967. doi:10.1177/03635465231151252
- Kon E, Di Matteo B, Verdonk P, et al. Aragonite-Based Scaffold for the Treatment of Joint Surface Lesions in Mild to Moderate Osteoarthritic Knees: Results of a 2-Year Multicenter Prospective Study. Am J Sports Med. 2021;49(3):588-598. doi:10.1177/0363546520981750
- Kon E, Filardo G, Shani J, et al. Osteochondral regeneration with a novel aragonite-hyaluronate biphasic scaffold: up to 12-month follow-up in a goat model. J Orthop Surg Res. 2015;10:81. Published 2015 May 28. doi:10.1186/s13018-015-0211-y
- Matta C, et al. Differentiation. 2019;107:24-34.
- Chubinskaya S, et al. Knee Surg Sports Traumatol Arthrosc. 2019;27(6):1953-1964.
About Smith+Nephew
Smith+Nephew is a portfolio medical technology business focused on the repair, regeneration, and replacement of soft and hard tissue. We exist to restore people’s bodies and their self-belief by using technology to take the limits off living. Our 17,000 employees deliver this mission every day, making a difference to patients’ lives through our product portfolio and innovations in Orthopaedics, Sports Medicine & ENT, and Advanced Wound Management.
Founded in Hull, UK, in 1856, we operate in around 100 countries, with 2024 sales of $5.8 billion. Smith+Nephew is a constituent of the FTSE100 (LSE:SN, NYSE:SNN). For more information, visit www.smith-nephew.com.